Govt proposes to drop repeat virus assessments on plasma-based medicines | India Information
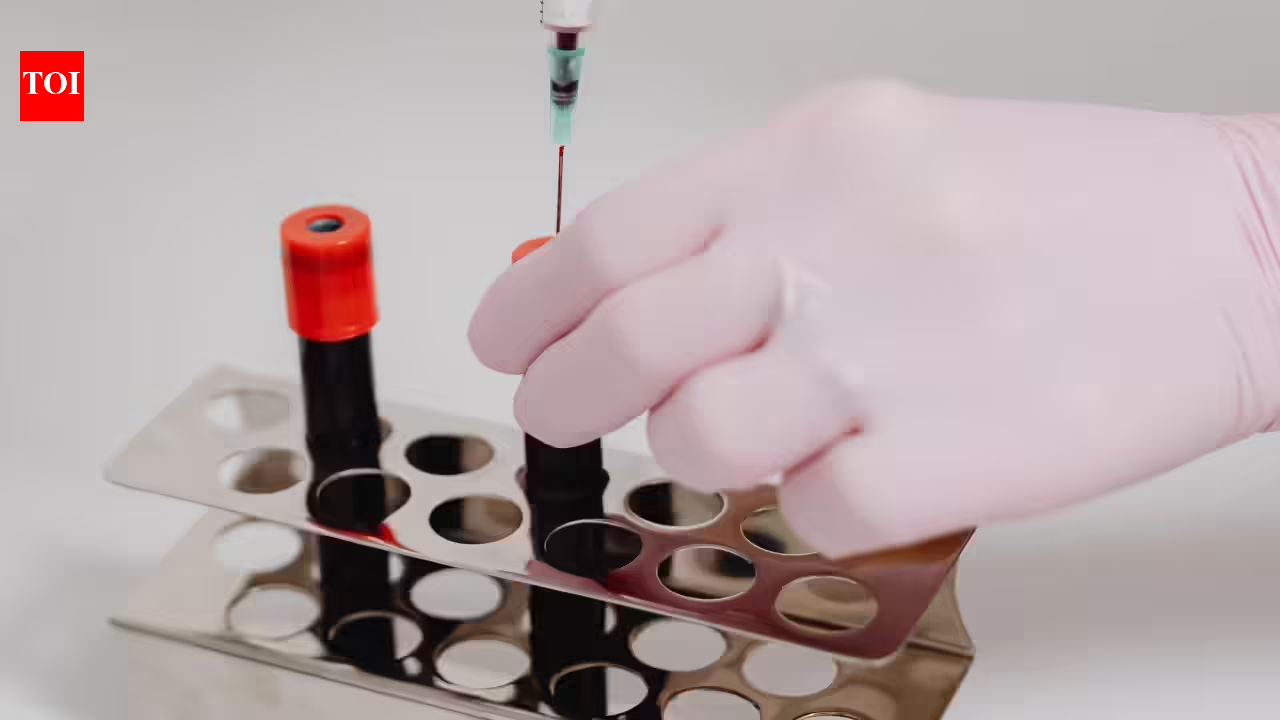
NEW DELHI: The Centre has proposed amending drug guidelines to cease repeating virus assessments on medicines created from human plasma, saying the uncooked plasma used to supply these merchandise is already screened for infections reminiscent of HIV and hepatitis earlier than manufacturing begins.These medicines embrace albumin, intravenous immunoglobulin (IVIG), and clotting components reminiscent of Issue VIII and Issue IX, that are used to deal with immune issues, extreme infections and bleeding situations reminiscent of haemophilia.Officers mentioned the transfer goals to align India’s drug laws with worldwide pharmacopoeia requirements. World pointers require pooled plasma to be examined for hepatitis B floor antigen, hepatitis C virus RNA and HIV antibodies earlier than it’s used for fractionation, and solely plasma that assessments destructive is cleared for manufacturing plasma-derived medicines.At the moment, plasma collected for manufacturing these medicines is first pooled and examined for viruses, together with HIV, hepatitis B and hepatitis C. Nevertheless, as soon as medicines are produced from this screened plasma, the completed merchandise are once more examined for a similar viral markers below present guidelines. The federal government now proposes to take away this second spherical of testing.The well being ministry has issued a draft notification searching for public feedback on amendments to the Medicine Guidelines, 1945, which regulate the testing of blood-derived merchandise. Dr Aseem Kumar Tiwari, Senior Director, Division of Transfusion Drugs, Medanta, Gurugram, mentioned that surplus plasma collected from blood donors could also be utilized by plasma fractionators to fabricate a number of life-saving medicines.“Plasma-derived medicinal merchandise (PDMP) reminiscent of albumin, intravenous immunoglobulin (IVIG), and clotting components like Issue VIII and Issue IX are extensively used to deal with immune issues, extreme infections and bleeding situations reminiscent of haemophilia,” he mentioned.He added that blood centres usually generate surplus plasma after assembly affected person wants, which may be equipped to specialised fractionation amenities the place totally different proteins are separated to fabricate these medicines.Dr Tiwari mentioned PDMPs bear a number of security checks earlier than reaching sufferers. “Donated plasma is screened for infections reminiscent of HIV, hepatitis B, hepatitis C, malaria, and syphilis, and the manufacturing course of consists of viral inactivation steps to make sure security,” he mentioned.“These medicines, identified globally as plasma-derived medicinal merchandise, haven’t been linked to an infection transmission due to stringent testing and viral inactivation throughout manufacturing,” he added.Officers say repeating the identical viral assessments on the completed product stage creates duplication not required below international practices. The proposed modification seeks to rationalise testing necessities whereas sustaining strict security checks on the plasma screening stage.The draft guidelines have been issued after session with the Medicine Technical Advisory Board, and stakeholders have been given 30 days to submit feedback earlier than the modification is finalised.